Metformin: A drug for Diabetes and friends with 2DG
Summary
Metformin is an antihyperglycemic drug, also used in the treatment of cancer nowadays. It has the ability to inhibit oxidative phosphorylation in cells. It also increases the expression of p53 in cells that induce apoptosis. If metformin is used in combination with 2DG, which is a glucose antimetabolite, it proves to be very effective in cancer therapeutics. The mechanisms of action of metformin in diabetes and cancer are explained in this article.
Metformin
It is an anti-hyperglycemic drug that is given to type-2 diabetes patients to control their Glucose levels in the body. It is a first-line therapy to cure type 2 diabetic patients. Metformin is used as an adjunct to diet and exercise.
This drug was first approved in 1972 in Canada and was later approved by FDA in the USA in 1995. In type 2 diabetic patients, it lowers the blood glucose levels without causing hypoglycemia in patients. It is commonly called an “insulin sensitizer” because of its role in lowering insulin resistance in the body. It is also very effective for weight loss in diabetic patients (Update et al., 2002).
Diabetes mellitus
Diabetes occurs when a person’s body is unable to take up glucose in the cells so that it can not be used for the production of energy. As a result, glucose gets accumulated in the bloodstream.
If diabetes is not controlled, it can result in serious consequences, like damaging different body organs including the heart, nerves, eyes, and kidneys (Malik & Andag-silva, 2014).
Cancer
It is a broad term representing a disorder that results from cellular mutations, and these mutations make the cell grow and proliferate unlimitedly. Many forms of cancer are visible, these are called tumors. While the invisible forms of cancer are also found like leukemia or blood cancer. All cells of the body have built in phenomenon for cell death called apoptosis. The body keeps on replacing the cells. But if cells get some mutations that inhibit the process of apoptosis in them, this leads to cancer.
Normal cells in the body undergo apoptosis that results from stimuli either internal or external. P53 protein is the main regulator of apoptosis, as it has the ability to regulate or to suppress the process of transcription in cells. More than 50% of tumors have the ability to lose p53, they also activate PI3/AKT signaling in order to evade apoptosis. Cancer cells also have the ability to produce IGF survival factors. Thus, cancer cells have many hallmarks that make them distinct from normal cells.
Metformin mechanism of action
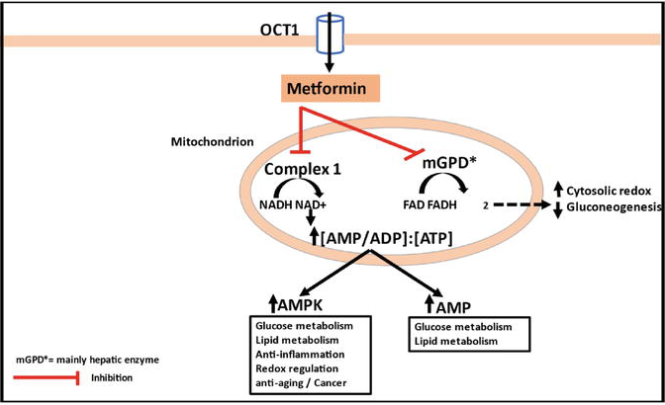
The mechanism of action of metformin is quite different from other antihyperglycemic drugs. It decreases gluconeogenesis by decreasing blood glucose levels. As a result, absorption of glucose by the intestine decreases, and the sensitivity of insulin increases due to increased uptake of glucose by peripheral tissues. It is found experimentally that metformin inhibits complex 1 of mitochondria, which decreases the blood glucose level causing a positive effect to control blood glucose level in type 2 diabetes patients.
When a patient ingests metformin, it is taken to hepatocytes by organic cation transporter-1 (OCT-1). Metformin is positively charged, so when it enters the cell, it gets accumulated in the cytosol and mitochondria because of negative membrane potential on the inner sides of the plasma membrane and mitochondrial membrane. It inhibits the mitochondrial complex 1 that decreases the production of ATP in cells so that ADP: ATP and AMP: ATP increase in the cytoplasm (Foretz et al., 2019).
As a result, AMP-activated Protein Kinase (AMPK) is activated in the cell, which is an important player in regulating the metabolism of glucose in the cell. This increased AMP in cells also inhibits the Fructose-1,6-bisphosphate enzyme, which ultimately inhibits the process of gluconeogenesis. This also inhibits the adenylate cyclase enzyme, that is involved in the production of cAMP. So, the production of cAMP in cells also decreases. cAMP plays an important role in cell signaling (Sahra et al., 2010).
The activated AMPK enzyme causes phosphorylation of acetyl CoA carboxylase so that the synthesis of fats is inhibited, causing fatty acid oxidation in the cell. This decreases the lipid stores in the liver, ultimately increasing insulin sensitivity.
In enterocytes, metformin increases anaerobic glycolysis, this decreases the uptake of glucose by the cells and starts lactic acid formation, which is delivered to the liver (Foretz et al., 2019).
Use of Metformin for the treatment of cancer
Many studies have proved that metformin can be given to cancer patients as it reduces the growth of tumors. It affects the metabolism in malignant cells and also inhibits the mTOR signaling pathways that are important for energy production in cells. mTOR is an important pathway regulating the differentiation of cells, transcription, and translation processes. So, if this pathway is inhibited, the survival and proliferation of cells become difficult.
In hepatocytes, as mentioned earlier, metformin blocks the respiratory chain complex 1. Thus, inhibiting the process of oxidative phosphorylation in cells. As cancer cells depend for their energy on two main processes, glycolysis, and oxidative phosphorylation. So, inhibiting mitochondrial complex-1 will make the cells deprived of energy (Morales & Morris, 2015).
It is also found experimentally that metformin enhances the expression levels of p53 in the cancer cells, which induces apoptosis. Thus, metformin has an anti-proliferative ability as well.
Combination of Metformin and 2DG
Cancer cells proliferate and metastasize rapidly. For this, they require a large amount of energy. This energy is produced by two main mechanisms in the cancer cells.
- Glycolysis
- Oxidative phosphorylation
If there is no energy production in the cell, it cannot survive. So, to stop the occurrence of cancer in a patient, we need to stop the production of energy in these malignant cells. So, we should inhibit the above-mentioned processes for energy production.
2DG is a glucose anti-metabolite having structural similarity with the glucose molecule, that binds the glucose transporters. It can enter the cell and accumulate there but cannot be metabolized like glucose for energy production. In short, it inhibits the glycolytic pathway in cells. Moreover, it also activates some protein enzymes like Caspases and PARP that activate the process of apoptosis in the cells.2DG also inhibits the glycosylation of different proteins that are important for signaling pathways in the cells. So, these proteins become non-functional in cells.
Metformin was commonly used as an antidiabetic drug for years. But recent studies have shown that it has anti-tumor activity as well. This drug reduces the growth of different tumor types. Moreover, it has the ability to induce apoptosis in the cell.
Experiments have shown that if 2DG and Metformin are given together to cancer patients, this will be a more effective therapy. 2DG stimulates the level of p53 in malignant cells, but metformin has the ability to increase the expression of p53 in cells and reduce the expression of BCl2 protein that is anti-apoptotic (Shafaee et al., 2019).
So, based on these findings, if 2DG and metformin are used in combination for tumor patients, they will block energy production by inhibiting both main mechanisms. 2DG inhibits glycolysis, while metformin inhibits oxidative phosphorylation. Moreover, both have the ability to induce the process of apoptosis in malignant cells by activating different proteins (Sahra et al., 2010).

Conclusion
Metformin is an antihyperglycemic drug that is used commonly to control blood sugar levels in type 2 diabetic patients. Diabetes mellitus is of different types, the most common being type 2 diabetes. The mechanism of action of metformin is different from that of other antihyperglycemic drugs. It acts in the liver, blocking the mitochondrial complex 1 in hepatocytes. It inhibits the process of oxidative phosphorylation in the cells it acts on.
It was found recently that metformin can also be used to cure tumor patients. As it has antiproliferative activity in malignant cells by inhibiting oxidative phosphorylation in them. Moreover, it also has the ability to induce apoptosis in malignant cells.
2DG is also an inhibitor of glycolysis being used for cancer treatment. It is proved experimentally that 2DG and metformin are used in combination. They will be able to act more effectively against tumor cells. This will not only stop the proliferation and metastasis in cancer cells but will also induce apoptosis in them.
References
Foretz, M., Guigas, B., & Viollet, B. (2019). Understanding the glucoregulatory mechanisms of metformin in type 2 diabetes mellitus. Nature Reviews Endocrinology, 15(10), 569–589. https://doi.org/10.1038/s41574-019-0242-2
Malik, R., & Andag-silva, A. (2014). General aspects of diabetes mellitus Related papers. Handbook OfClinical Neurology, Vol. 126 (3rd Series), 211–222.
Morales, D. R., & Morris, A. D. (2015). Metformin in cancer treatment and prevention. Annual Review of Medicine, 66(November 2014), 17–29. https://doi.org/10.1146/annurev-med-062613-093128
Sahra, I. Ben, Tanti, J. F., & Bost, F. (2010). The combination of metformin and 2-deoxyglucose inhibits autophagy and induces AMPK-dependent apoptosis in prostate cancer cells. Autophagy, 6(5), 670–671. https://doi.org/10.4161/auto.6.5.12434
Shafaee, A., Pirayesh Islamian, J., Zarei, D., Mohammadi, M., Nejati-Koshki, K., Farajollahi, A., Aghamiri, S. M. R., Rahmati Yamchi, M., Baradaran, B., & Asghari Jafarabadi, M. (2019). Induction of Apoptosis by a Combination of 2-Deoxyglucose and Metformin in Esophageal Squamous Cell Carcinoma by Targeting Cancer Cell Metabolism. Iranian Journal of Medical Sciences, 44(2), 99–107.
Update, M. A., Kirpichnikov, D., Mcfarlane, S. I., & Sowers, J. R. (2002). Update OF OF. 24.
